In the 1986 horror movie, “The Fly”, a quantum teleportation accident slowly merges eccentric scientist Dr. Seth Brundle (Jeff Goldblum) and a common house fly together into a being known as “Brundlefly”. Over the course of the movie, Dr. Brundle becomes more grotesque as he concurrently loses his humanity. Since teleportation is not real, we don’t need to worry about the dangers of fly-human hybrids. However, fruit flies are one of the most common animals used in biological experiments, and as a result we know of quite a few very strange mutations. Here are my top 5 coolest fly mutants.

5. Hedgehog
The hedgehog gene plays a number of important roles in fruit fly development. The gene is very active early in development when the body segments are being organized.1 When the gene is mutated so that it no longer works, the fruit fly embryo develops strange, spiny projections all around its body (hence the name, hedgehog). A version of this gene that is found in vertebrates was subsequently named sonic hedgehog.
4. Tinman
The Tinman gene is one of the absolute earliest genes involved in heart development in fruit flies.2 The gene was named after the “Wizard of Oz” character, the Tinman, because when it is mutated, the fruit fly completely fails to develop a heart. Unlike the Wizard of Oz character, flies can’t live without hearts, so this mutation is absolutely lethal.
3. Argos
The argos gene plays a role in the development in fruit fly eyes. Specifically, it helps organize which cells should play which roles in the eye. When the argos gene is overactive, it creates a “rough eye” phenotype on the eye, where abnormal bulges begin to form.3

2. Ultrabithorax
Your average fruit fly has one set of wings, and right behind them, a set of small structures called halteres that are used for balance during flight. The development of these halteres is controlled largely by a gene called ultrabithorax.4 When this gene is mutated and stops functioning, the fly instead develops the previous segment of its body a second time. This results in a “bithorax fly”, which contains an unusual second entire set of wings.

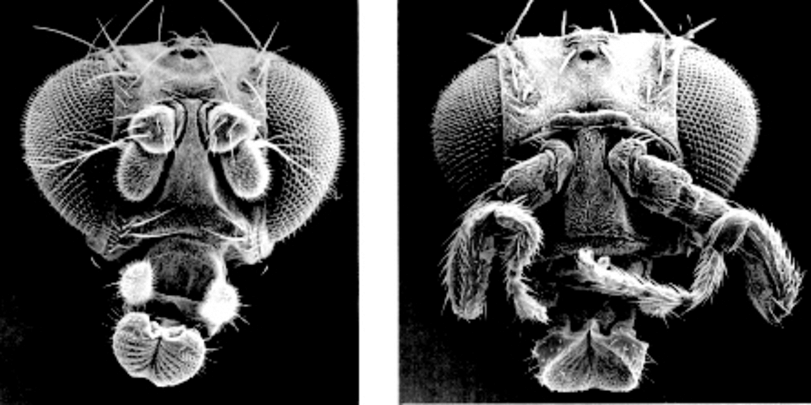
1. Antennapedia
The antennapedia gene can do a couple of crazy things. In normal circumstances, this gene controls where the first pair of legs will develop in the fruit fly.5 When the gene is mutated to stop functioning, the previous appendage develops in place of the first legs. This means that the fly will have antennae growing where legs should be. But that’s not even the craziest thing that can be done with antennapedia. If the gene is overexpressed into the head segment, legs will grow in place of antennae! This is one of the coolest examples of real-life science fiction that I can think of, which is why antennapedia lands the #1 spot on this list.
References:
1. Nüsslein-Volhard, Christiane, and Eric Wieschaus. “Mutations Affecting Segment Number and Polarity in Drosophila.” Nature, vol. 287, no. 5785, 1980, pp. 795–801., doi:10.1038/287795a0.
2. Bodmer, Rolf. “The Gene Tinman Is Required for Specification of the Heart and Visceral Muscles in Drosophila.” Development, vol. 118, no. 3, 1993, pp. 719–729.
3. Wemmer, T, and C Klämbt. “A Genetic Analysis of the Drosophila Closely Linked Interacting Genes Bulge, Argos and Soba.” Genetics, vol. 140, no. 2, 1995, pp. 629–641., doi:10.1093/genetics/140.2.629.
4. Weatherbee, S. D., et al. “Ultrabithorax Regulates Genes at Several Levels of the Wing-Patterning Hierarchy to Shape the Development of the Drosophila Haltere.” Genes & Development, vol. 12, no. 10, 1998, pp. 1474–1482., doi:10.1101/gad.12.10.1474.
5. Schneuwly, Stephan, et al. “Redesigning the Body Plan of Drosophila by Ectopic Expression of the Homoeotic Gene Antennapedia.” Nature, vol. 325, no. 6107, 1987, pp. 816–818., doi:10.1038/325816a0.



